
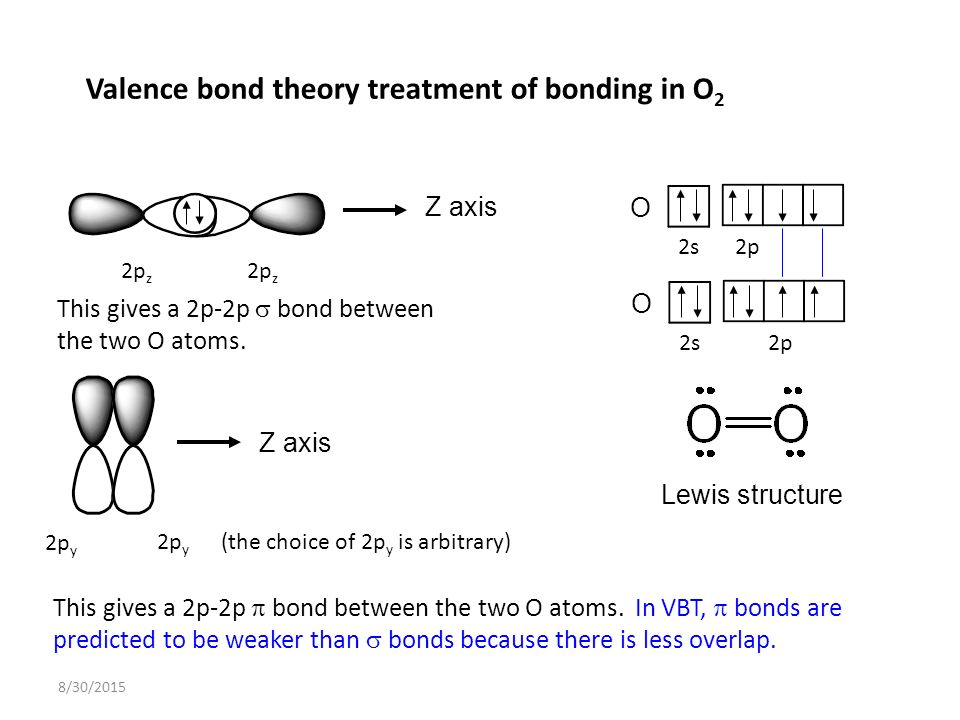
Here, side by side overlapping takes place. ”Pi bonds are the result of the pairing of unbound p-orbital electrons between two atoms. When two fluorine atoms having opposite spins approach each other, their p orbitals overlap with each other. Here, the electronic configuration of fluorine is F: 1s 2 2s 2 2px 2 2py 2 2pz 1. The formation of fluorine (F 2) molecules is an example of the p-p sigma bond.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
